How To Make A Model Of The Molecular Structure Of Water
Water is the most-studied molecule in all of science. It is a simple molecule, consisting of just one oxygen atom and two hydrogen atoms. It is one of the easiest atoms to build a model of, and is therefore an excellent starting point for students learning to build molecular models.
Step 1
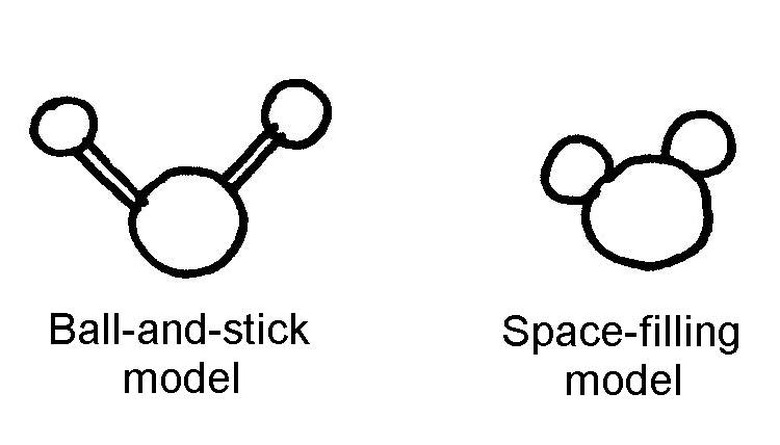
Decide whether you want to make a ball-and-stick model or a space-filling model. Both are used in science books and represent different ways of showing the chemical bonds between the atoms.
Step 2
Make the ball-and-stick model by using three candies and two toothpicks. Choose two colors: one color to represent the oxygen atom and one color to represent the two hydrogen atoms. Push the toothpicks into the candy far enough so that they don't fall off.
Step 3
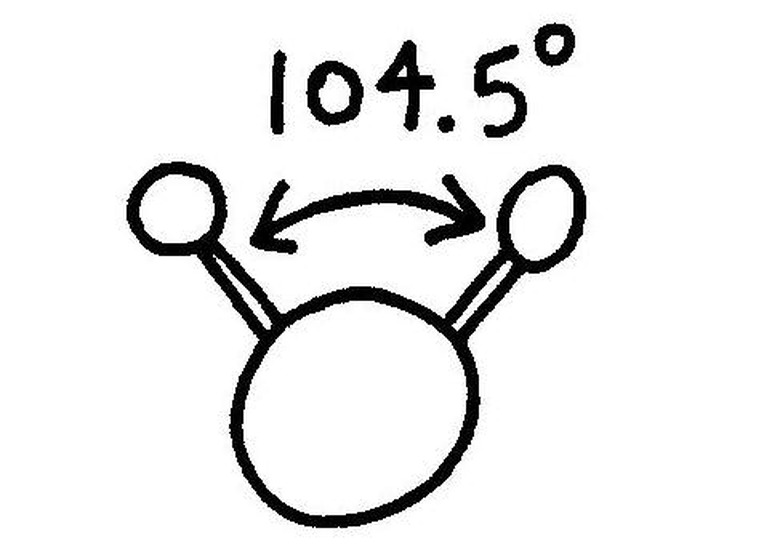
Use a protractor to measure the angle between the toothpicks if you would like to add extra accuracy to your model. The angle between the hydrogen atoms in a water molecule is 104.5 degrees.
Step 4
Make a space-filling molecule by breaking a toothpick in half and using the halves to stick the candies together so that they touch (if the halves are still too long, you can make them even smaller). You might want to use smaller candies for the hydrogen atoms since, in reality, hydrogen atoms are smaller than oxygen atoms.
Things Needed
- Toothpicks
- Candies or marshmallows
- Optional: protractor
TL;DR (Too Long; Didn't Read)
Consider using healthy snack items for this craft, especially if you will be making and eating a lot of them. You could use grapes, raisins, dried fruits or pieces of cheese or carrot.
Cite This Article
MLA
RuthJ, . "How To Make A Model Of The Molecular Structure Of Water" sciencing.com, https://www.sciencing.com/make-model-molecular-structure-water-4487842/. 24 April 2017.
APA
RuthJ, . (2017, April 24). How To Make A Model Of The Molecular Structure Of Water. sciencing.com. Retrieved from https://www.sciencing.com/make-model-molecular-structure-water-4487842/
Chicago
RuthJ, . How To Make A Model Of The Molecular Structure Of Water last modified August 30, 2022. https://www.sciencing.com/make-model-molecular-structure-water-4487842/