What Is Anhydrous Diethyl Ether?
Ethers are a class of organic compounds that contain an ether functional group. Ether has a general formula of R-O-R', where R and R' represent any alkyl or aryl group attached to the oxygen atom. Diethyl ether, or ethyl ether, is one example of ether.
However, due to its prevalence in organic chemistry and biochemistry, diethyl ether is often used interchangeably with "ether." By removing water from diethyl ether, it generates anhydrous (dry) diethyl ether, which is typically used as a solvent in organic chemistry labs.
Properties of Diethyl Ether
Properties of
Diethyl Ether
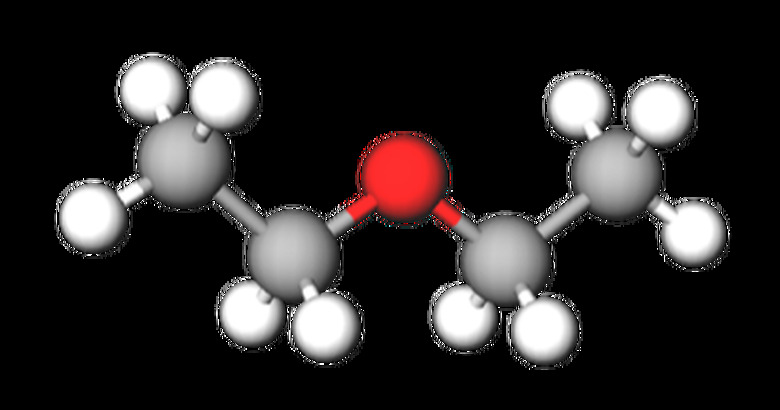
Diethyl ether has a chemical formula of C4H10O and a molecular mass of 74.12 g/mol. It is colorless but has a strong, distinct smell and a hot, sweet taste. Ethyl ether is highly flammable and volatile. The diethyl ether boiling point is 34.6 °C, and the diethyl ether melting point is −116.3 °C. The structure of diethyl ether is CH3-CH2-O-CH2-CH3, with the ball-and-stick model as follows:
Although ethers have high chemical stability, they can undergo cleavage with certain reagents or under extreme conditions. Diethyl ether can also form peroxides in the presence of oxygen and light. It is also a Lewis and Brønsted base and an important solvent for Grignard reagents.
Synthesis of Diethyl Ether
Synthesis of Diethyl Ether
Industrial production of diethyl ether is accomplished by the condensation of alcohols. At elevated temperatures (130°C to 140 °C), two molecules of alcohol condense to form one molecule of ether and water. This reaction is catalyzed by acids, usually sulfuric acid, with the mechanism depicted below:
At temperatures higher than 150 °C, however, ethanol undergoes dehydration (elimination) to form ethylene. This condensation method is also unsuitable for making unsymmetrical ethers. Since there is no way to control which OH gets protonated or acts as a nucleophile, it will result in a mixture of two different symmetrical and the desired unsymmetrical ether.
For anhydrous diethyl ether, the product from the condensation is dried over thin slices of metallic sodium followed by distillation in a water bath. A more convenient method of drying laboratory diethyl ether is to use a desiccant such as molecular sieves, which are interconnecting networks of alumina and silica tetrahedra.
It can be activated by heating to generate uniform cavities which selectively absorb molecules of a specific size. For diethyl ether, a pore size of 4Å is appropriate for the removal of water.
Uses of Diethyl Ether
Uses of Diethyl Ether
Ether was used throughout medical history as a treatment for scurvy or pulmonary inflammation. In 1846, an American dentist William T. G. Morton, for the first time, demonstrated in public the use of ether as an effective surgical anesthetic. The use of diethyl ether has great advantages over chloroform because of its larger therapeutic/safety window, but it still has undesirable side effects.
Today, ethers have been replaced by modern anesthetic agents with even better properties. Diethyl ether is still a common laboratory solvent for chemical reactions and liquid-liquid extraction. For instance, it is used as a solvent in the production of cellulose plastics. Due to its low flash point, it is also used as a starting fluid for certain engines in cold climates.
Diethyl Ether Hazards
Diethyl Ether Hazards
Diethyl ether hazards, when inhaled, include nausea, vomiting and even loss of consciousness. It can also cause irritation and burns when in direct contact with eyes and skin.
Ethyl ether is also extremely flammable and can be ignited by flame, heat and static electricity. With air and light, ether tends to form explosive peroxides, which is particularly dangerous when evaporation to dryness is attempted.
As a result, commercial diethyl ether is often stabilized with butylated hydroxytoluene (BHT) to reduce the formation of peroxides. Peroxide test strips are also available for measuring the peroxide content in old bottles of ether in the lab.
Cite This Article
MLA
Luo, Lan. "What Is Anhydrous Diethyl Ether?" sciencing.com, https://www.sciencing.com/anhydrous-diethyl-ether-6025498/. 26 March 2020.
APA
Luo, Lan. (2020, March 26). What Is Anhydrous Diethyl Ether?. sciencing.com. Retrieved from https://www.sciencing.com/anhydrous-diethyl-ether-6025498/
Chicago
Luo, Lan. What Is Anhydrous Diethyl Ether? last modified August 30, 2022. https://www.sciencing.com/anhydrous-diethyl-ether-6025498/