How To Build A Clorox Bleach Battery
Batteries can be constructed using common household materials. It's all a matter of chemistry: When acids are present in a solution, ions are produced. When two dissimilar metals are introduced into the solution, an electric current forms between them, producing electricity. Create a bleach battery the next time you need a science fair project.
Step 1
Fill a plastic or other nonmetallic cup about 2/3 to 3/4 full with tap water. Add the following: 1 teaspoon of salt, 1 teaspoon of vinegar and about 1 teaspoon of household bleach (Clorox Bleach works well).
Step 2
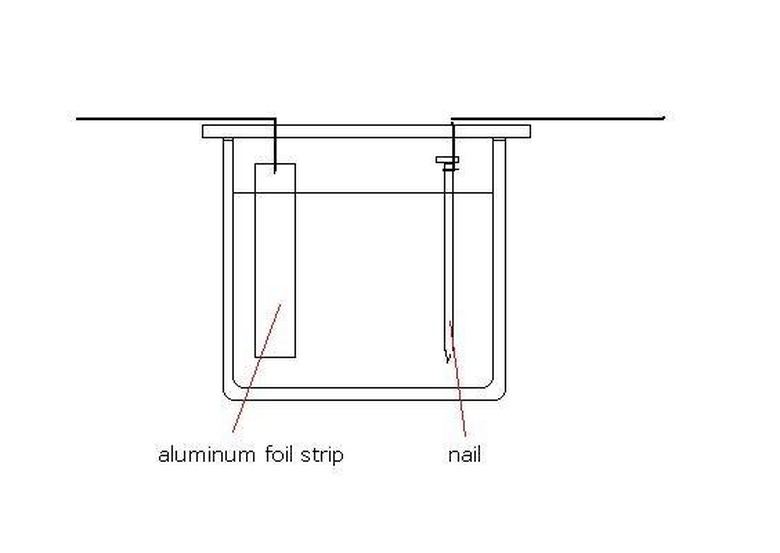
Place a pencil or small dowel rod on the rim of the glass so that it makes a "bridge" that extends over the top of the cup.
Step 3
Take two 12- to 18-inch pieces of insulated wire (20-gauge works well) and use wire strippers to remove about 1 inch of insulation from both ends of the wires.
Step 4
Take one wire and wrap the bare end around the head end of a nail. Wrap the wire around the pencil so that the nail is suspended in the bleach solution.
Step 5
Wrap the second wire onto a strip of aluminum foil. Then wrap the wire around the pencil so that the foil strip is suspended in the bleach solution.
Step 6
Your battery is complete. To measure the voltage output, connect the wire ends to a multimeter. The output will be low, but in some cases, you may be able to power a small device with this battery or a series of interconnected batteries.
Things Needed
- Plastic cup
- Water
- Salt
- Vinegar
- Clorox bleach (or other household chlorine bleach)
- Pencil
- Two 12- to 18-inch pieces of 20-gauge insulated wire
- Wire strippers
- Aluminum foil
- Uncoated iron nail
- Multimeter
TL;DR (Too Long; Didn't Read)
Try different metals in the bleach solution to see what difference you get in electricity output.
Cite This Article
MLA
Liandi, Nichole. "How To Build A Clorox Bleach Battery" sciencing.com, https://www.sciencing.com/build-clorox-bleach-battery-5316693/. 24 April 2017.
APA
Liandi, Nichole. (2017, April 24). How To Build A Clorox Bleach Battery. sciencing.com. Retrieved from https://www.sciencing.com/build-clorox-bleach-battery-5316693/
Chicago
Liandi, Nichole. How To Build A Clorox Bleach Battery last modified August 30, 2022. https://www.sciencing.com/build-clorox-bleach-battery-5316693/