How To Calculate How Many Rings In An Atom
In order to calculate how many rings are in an atom, you need to know how many electrons the atom has. The rings, also known as electron shells, can hold a variable amount of electrons depending on its shell number. For example, the first shell can hold only two electrons. If the atom has more than two electrons, then that atom must have more than one ring. To determine how many electrons a shell can hold, you use a formula that calculates the number of electrons possible in a given shell. You need to fill one shell, starting with shell number one, before filling another. The last shell does not have to be full of electrons.
Step 1
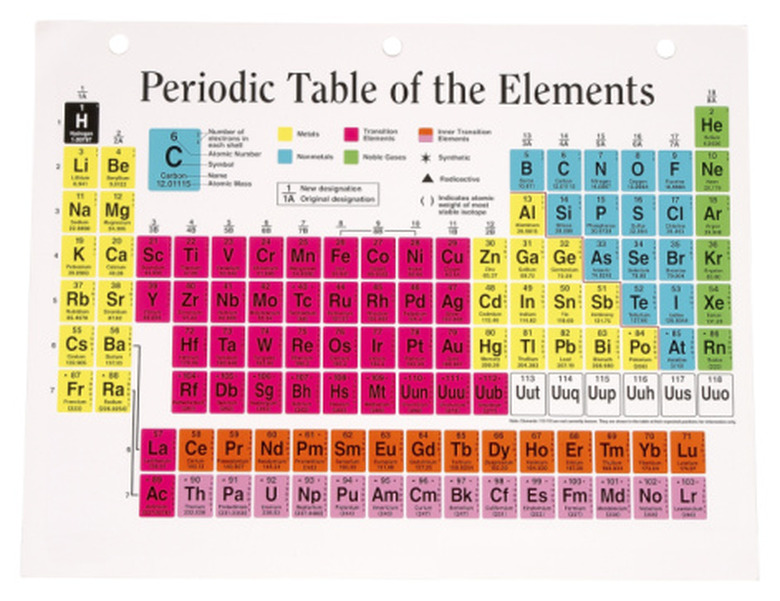
Find the number of electrons in the atom by using the periodic table. The number of electrons is equal to the atom's atomic number, which is at the top left of the element. For example, assume you want to know how many rings are in the element neon. Neon on the periodic table has an atomic number of 10, so it has 10 electrons.
Step 2
Square the ring number, then multiply the result by two. Check to see if the ring is full or not. If the ring is full, then move to the next ring. If the ring is not full, then that is how many rings are needed. Starting with the first ring, 1 squared = 1; and 1 x 2 = 2, so that is the maximum number of electrons that ring one can hold. Subtract this from your element's atomic number. Still using neon as an example, you now have eight remaining electrons.
Step 3
Calculate the number of electrons in the next ring. Using the formula, 2 squared = 4; and 4 x 2 = 8, which indicates that the second ring can hold a maximum of eight electrons. In our example, we had eight remaining electrons, so this ring is full and no electrons remain. Therefore, an atom of neon has two rings.
Cite This Article
MLA
McBride, Carter. "How To Calculate How Many Rings In An Atom" sciencing.com, https://www.sciencing.com/calculate-many-rings-atom-8550529/. 24 April 2017.
APA
McBride, Carter. (2017, April 24). How To Calculate How Many Rings In An Atom. sciencing.com. Retrieved from https://www.sciencing.com/calculate-many-rings-atom-8550529/
Chicago
McBride, Carter. How To Calculate How Many Rings In An Atom last modified March 24, 2022. https://www.sciencing.com/calculate-many-rings-atom-8550529/