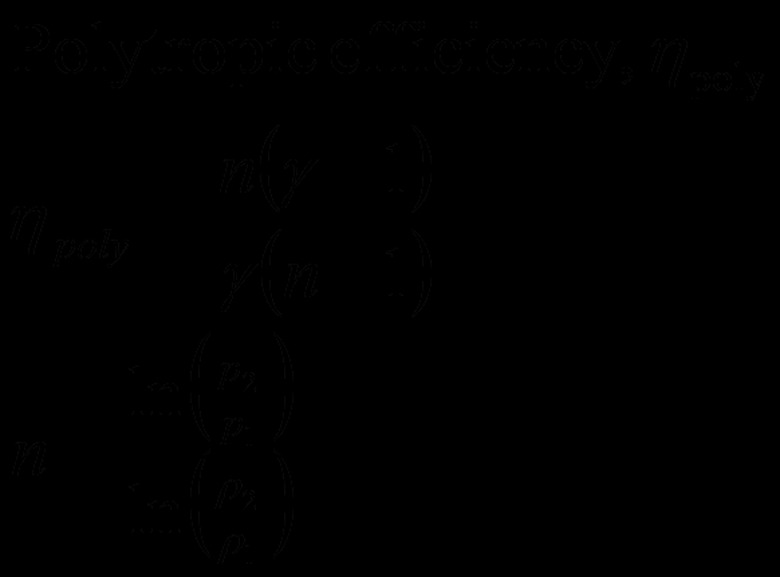How To Calculate Polytropic Efficiency
Polytropic efficiency is a value used to describe the efficiency of a compressor. A polytropic process is more difficult to analyze than a system under the isentropic or adiabatic assumptions. The difficulty arises from the fact that heat flows in and out of the system, and this added energy changes some of the basic gas properties, specifically the ratio of specific heats. For air, this value is a constant equal to 1.4. For a polytropic process, each new calculation requires a new value for this ratio.
Step 1
Obtain the pressure ratio and density ration across the compressor stage you are measuring. These can come from measurements taken at inlet and outlet positions, or you can calculate them based on your compressor's design characteristics. These values are from compressibility tables, just to illustrate the process: p2/p1 = 4.5 rho2/rho1 = 2.667
Step 2
Calculate the value for the polytropic ration of specific heats by dividing the natural logarithm of the pressure ratio by the natural logarithm of the density ratio. In this example, "n" represent this ratio. n = 1.504077/0.980954 n = 1.533927
Step 3
Determine the value for the ratio of specific heats for the gas you are using in your compressor. For air, the value is gamma = 1.4.
Step 4
Use "n" and gamma to calculate the polytropic efficiency, or eta. eta = (n(gamma – 1))/(gamma(n – 1)) eta = (1.533927_0.4)/(1.4_0.533927) eta = 0.821
Step 5
Convert the polytropic efficiency value to a percentage for reporting purposes, or leave it as is if you intend to use it in further calculations. eta = 0.821*100 eta = 82.1 percent
TL;DR (Too Long; Didn't Read)
Keep as many decimal places as practical throughout all of the calculations in order to maintain a high degree of accuracy.
Cite This Article
MLA
McKenzie, Grant D.. "How To Calculate Polytropic Efficiency" sciencing.com, https://www.sciencing.com/calculate-polytropic-efficiency-5393724/. 24 April 2017.
APA
McKenzie, Grant D.. (2017, April 24). How To Calculate Polytropic Efficiency. sciencing.com. Retrieved from https://www.sciencing.com/calculate-polytropic-efficiency-5393724/
Chicago
McKenzie, Grant D.. How To Calculate Polytropic Efficiency last modified March 24, 2022. https://www.sciencing.com/calculate-polytropic-efficiency-5393724/