How To Figure Out The Chemical Symbol For Ions
An atom that has an equal number of protons and electrons is neither positive nor negative — it has no net charge. If that atom gains or loses electrons, however, it may become a cation, an ion with a positive charge, or an anion, an ion with a negative charge. Chemists use a very simple notation to represent ions in chemical reactions. Although you may need to remember some common polyatomic ions, for the most part, you can figure out the symbols for ions just using the periodic table.
Step 1
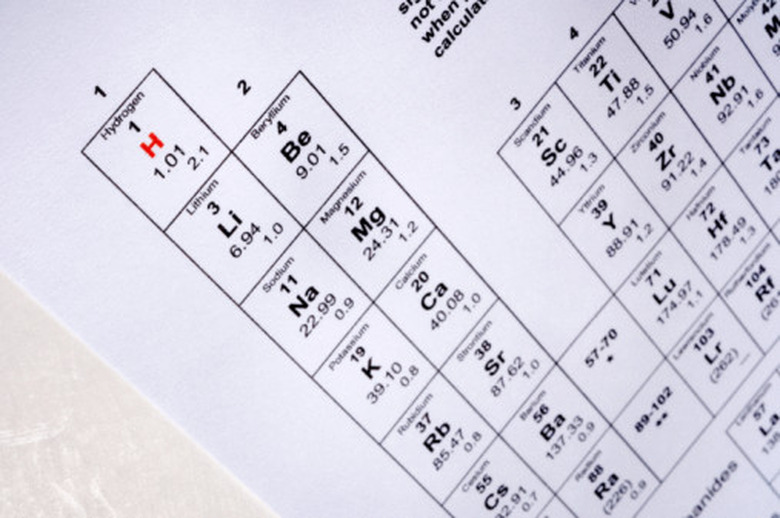
Determine whether the ion contains only a single element. If so, find the element that has been ionized on the periodic table. Sodium is in the first column, for example, while calcium is in the second.
Step 2
Write down the one- or two-letter symbol for the element from the periodic table. The symbol for sodium, for example, is Na, while the symbol for calcium is Ca.
Step 3
Determine how many electrons the atom has lost or gained. Elements in column 1 of the periodic table (e.g., sodium and potassium) will lose an electron when they react, while elements in the second column (e.g., calcium, magnesium and strontium) will generally lose two electrons when they react. Elements in group 17, the halogens (fluorine, chlorine, bromine and iodine) nearly always form ions that have gained a single electron. Sulfur and oxygen can form ions with a -2 charge. Elements in the middle of the table — the so-called transition metals — can lose a variable number of electrons. The number of electrons the transition metal atom has lost will be specified using Roman numerals after its name. Iron (III), for example, has lost three electrons, while iron (II) has lost two.
Step 4
Write a negative sign as a superscript, followed by the number of electrons the atom has gained OR write a positive sign as a superscript, followed by the number of electrons it has lost.
Example: The calcium ion would be written as Ca+2 (with the +2 as a superscript).
Step 5
Determine whether the ion contains more than one element (e.g., the sulfate ion). If so, look up its name in the table under the Resources section below. Each name has a symbol that corresponds to it. Sulfate, for example, is SO4 -2 (with the -2 as a superscript and the 4 as a subscript, since there are 4 oxygen atoms).
Things Needed
- Periodic table
- Pencil
- Paper
- Chemical formula
Cite This Article
MLA
Brennan, John. "How To Figure Out The Chemical Symbol For Ions" sciencing.com, https://www.sciencing.com/figure-out-chemical-symbol-ions-8257311/. 24 April 2017.
APA
Brennan, John. (2017, April 24). How To Figure Out The Chemical Symbol For Ions. sciencing.com. Retrieved from https://www.sciencing.com/figure-out-chemical-symbol-ions-8257311/
Chicago
Brennan, John. How To Figure Out The Chemical Symbol For Ions last modified August 30, 2022. https://www.sciencing.com/figure-out-chemical-symbol-ions-8257311/