How Are Isotopes Important In Studying The Human Body?
Isotopes are atoms of the same element that have different numbers of neutrons in their nuclei; when introduced into the human body, they can be detected by radiation or other means. The isotopes, used in conjunction with sophisticated equipment, give medical professionals a powerful "window" into the body, allowing them to diagnose diseases, study biological processes and investigate the movement and metabolism of drugs in living people.
Stable and Unstable Isotopes
Stable and Unstable Isotopes
Isotopes may be stable or unstable; the unstable ones emit radiation, and the stable ones do not. For example, the stable carbon-12 atom makes up 98.9 percent of all carbon on Earth; because the rarer carbon-14 isotope is radioactive and changes over time, scientists use it to determine the age of sometimes ancient biological specimens and materials. Chemically, stable and unstable isotopes act much the same, allowing doctors to substitute radioactive atoms for stable ones in drugs used to trace biological activities. Stable isotopes, easily identified with a device called a mass spectrometer, help researchers determine conditions in blood and tissue when radioactivity is not desirable.
Nutrition Research
Nutrition Research
Stable isotopes help nutrition scientists monitor the movement of minerals through the body. For example, of the four stable isotopes for iron, iron-56 naturally accounts for about 92 percent, and the rarest is iron-58 at 0.3 percent. A scientist gives a test subject doses of iron-58 and monitors the amounts of different iron isotopes in blood and other biological samples. Because iron-58 is heavier than iron-56, a mass spectrometer distinguishes them easily. Early samples will show more iron-56, but over time, iron-58 will be found in significant amounts in various tissues and substances, allowing the scientist to accurately measure how the subject's body processes iron.
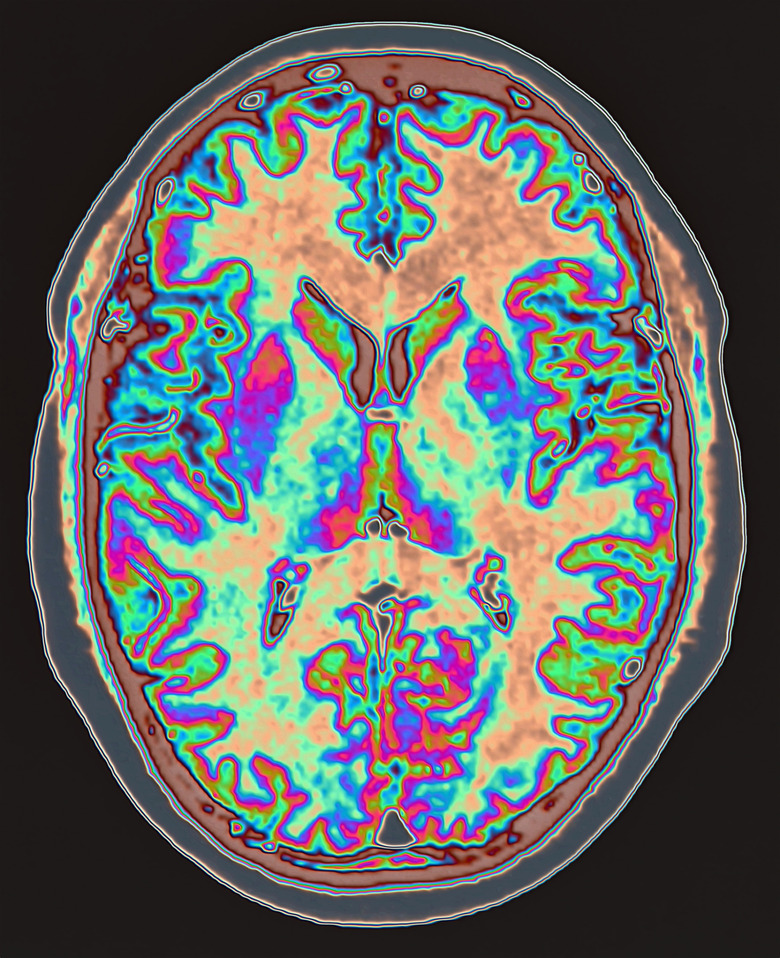
PET Scans
PET Scans
Positron Emission Tomography produces three-dimensional images of organs and tissues through the use of radioactive isotopes. The isotopes, such as fluorine-18, give off gamma radiation — a form of energy that passes through the body and into a detector. When combined with sugar and given to a patient, the fluorine migrates to those tissues that are actively metabolizing sugar, such as areas of the brain in a person working on math problems. PET scans show these body parts in clear detail. By observing the different levels of metabolism, a doctor can identify tell-tale signs of abnormalities such as tumors and dementia.
MPI Scans
MPI Scans
A Myocardial Perfusion Imaging scan uses radioactive isotopes to produce images in a method similar to a PET scan, but for monitoring the heart in real time. According to Stanford University Hospital, the technique employs isotopes such as technetium-99 or thallium-201. These isotopes are injected into a vein and find their way to the heart. A specialized camera picks up the emitted gamma rays and produce an image of the beating heart under resting and stress conditions, enabling a doctor to evaluate the organ's health.
References
- Columbia University: Carbon Isotopes
- Trace Sciences International: Stable Isotopes in Nutritional Studies
- Trace Sciences International: Stable Iron Isotopes
- Stanford Hospital: Myocardial Perfusion Scan, Resting and Stress
- National Institutes for Health: Dual Isotope High-Speed Myocardial Perfusion Imaging.
Cite This Article
MLA
Papiewski, John. "How Are Isotopes Important In Studying The Human Body?" sciencing.com, https://www.sciencing.com/isotopes-important-studying-human-body-14726/. 24 April 2017.
APA
Papiewski, John. (2017, April 24). How Are Isotopes Important In Studying The Human Body?. sciencing.com. Retrieved from https://www.sciencing.com/isotopes-important-studying-human-body-14726/
Chicago
Papiewski, John. How Are Isotopes Important In Studying The Human Body? last modified August 30, 2022. https://www.sciencing.com/isotopes-important-studying-human-body-14726/