How To Find The Number Of Neutrons In An Isotope
Atoms make up all matter. Everything with mass and volume, big or small, contains atoms. Amazingly enough, the tiny atoms contain even smaller particles. Protons, neutrons and electrons are the three main components of all atoms. The number and arrangement of these three unimaginably small particles determines the properties and behavior of the atoms that contain them.
TL;DR (Too Long; Didn't Read)
To find the number of neutrons in an isotope, subtract the number of protons from the atomic mass of the isotope. The atomic number of the element equals the number of protons. Calculating the number of neutrons then becomes atomic mass of the isotope minus the atomic number of the element equals the number of neutrons. For uranium-235, atomic number 92, the number of neutrons is 235-92=143, or 143 neutrons.
Particles in Atoms
Particles in Atoms
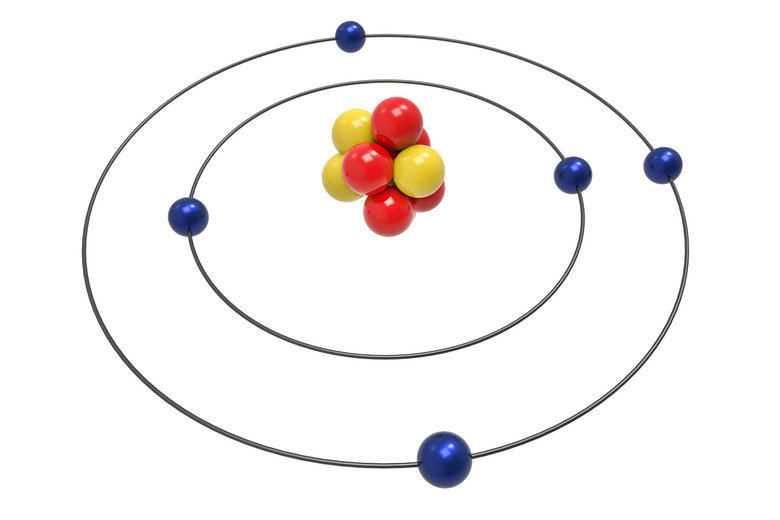
Almost all atoms contain three main particles: protons, neutrons and electrons. Protons and neutrons make up the nucleus, or center, of the atom. Electrons, much smaller than the protons and neutrons, circle the nucleus, zipping around at the speed of light. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. In a neutral atom, the number of protons equals the number of electrons, but the number of neutrons doesn't always equal the number of protons.
Identifying Atoms
Identifying Atoms
The number of protons in an atom determines which kind of element the atom forms. Hydrogen, first element on the periodic table, has only one proton. Helium, second on the periodic table, has two protons. Gold, number 79 on the periodic table, has 79 protons. The Periodic Table of Elements shows the elements in order of the number of protons in the atoms.
Isotopes of Atoms
Isotopes of Atoms
An isotope of an element means atoms with the same atomic number but different mass numbers. So, isotopes of an element have a different number of neutrons. Hydrogen has three isotopes. Hydrogen, the most common form of the atom, has one proton and one electron. Deuterium, one isotope of hydrogen, still only has one proton and one electron, but also has one neutron. Tritium, another isotope of hydrogen, still only has one proton and one electron but has two neutrons.
Calculating Neutrons
Calculating Neutrons
The mass of an atom consists of the combined masses of the protons and neutrons. The mass of the electrons is negligible in the overall mass of the atom. Protons measure about one atomic mass unit and neutrons measure only slightly more than one atomic mass unit. To find the number of neutrons in the atom, subtract the atomic number from the atomic mass.
The atomic number and the average atomic mass can be found on the periodic table. The mass of different isotopes is often written as part of the name of the isotope, however. Uranium-235 means the element uranium, atomic number 92, has 92 protons and an atomic mass of 235. On the other hand, uranium-238 has a mass of 238 but still has only 92 protons. An alternate method of writing an isotope shows the atomic mass as a superscript and the atomic number as a subscript. Uranium-235 could also be written as 23592 U where U is the standard abbreviation for uranium.
Using the hydrogen isotopes as examples, the "normal" atom of hydrogen has an atomic mass of 1, and the atomic number is 1, meaning the atom only has one proton. Using the formula, the atomic mass of 1 minus the atomic number, or number of protons, of 1 gives the equation 1-1=0, so the hydrogen atom has 0 neutrons. On the other hand, tritium, an isotope of hydrogen, has an atomic mass of 3 but the atomic number of hydrogen remains 1 because the atom only has one proton. Using the equation, atomic mass minus atomic number equals the number of neutrons, gives 3-1=2, so tritium has 2 neutrons.
Another common element, carbon, also has several isotopes. The normal carbon atom, atomic number 6, has an atomic mass of 12. Using the formula, atomic mass minus atomic number equals number of neutrons, shows 12-6=6, so the carbon-12 atom has 6 neutrons. Carbon-14, used for radioactive age dating of fossils under 10,000 years old, still has 6 protons but has an atomic mass of 14. Calculating the number of neutrons uses the same formula, so 14-6=8, so carbon-14 has 8 neutrons in its nucleus.
Cite This Article
MLA
Blaettler, Karen G. "How To Find The Number Of Neutrons In An Isotope" sciencing.com, https://www.sciencing.com/number-neutrons-isotope-8343646/. 10 May 2018.
APA
Blaettler, Karen G. (2018, May 10). How To Find The Number Of Neutrons In An Isotope. sciencing.com. Retrieved from https://www.sciencing.com/number-neutrons-isotope-8343646/
Chicago
Blaettler, Karen G. How To Find The Number Of Neutrons In An Isotope last modified March 24, 2022. https://www.sciencing.com/number-neutrons-isotope-8343646/