How Is PH Paper Made?
Infused with Indicators
Infused with Indicators
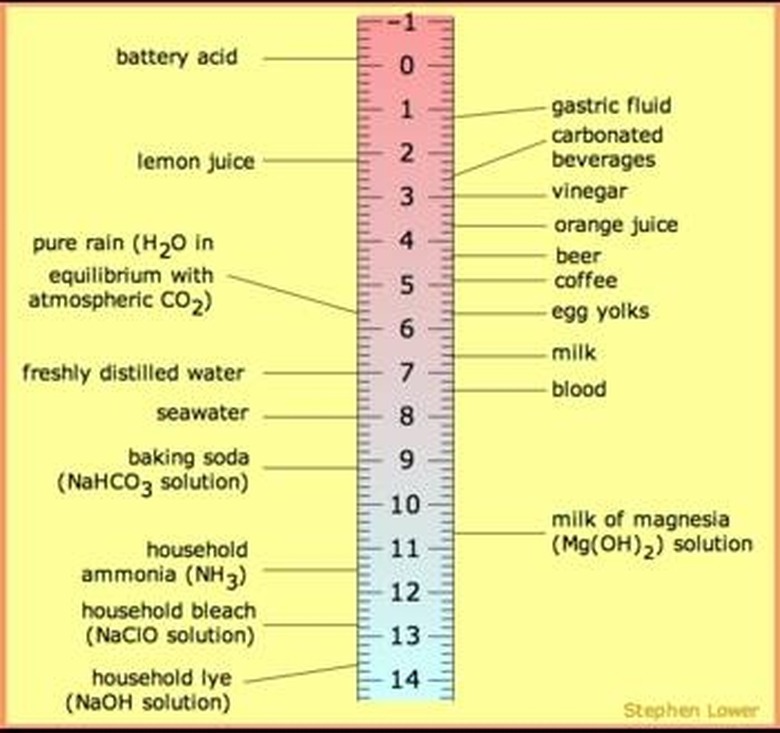
PH papers are paper infused, or saturated, with pH indicators or more frequently, a mixture of indicators. They are used to test for alkaline or acidity of a solution or substance such as soil. pH papers are usually sold as strips. These strips of paper (or sometimes fabric) change color when they come in contact with the material or solution being tested. To use it, you immerse it in the material or solution, wait for the number of seconds listed on the instructions, remove the paper and compare the color change to a scale. The scale will tell you if the substance is acidic or alkaline, and to what degree.
The indicators infused into the paper are predominantly weak acids. These acids react by changing color when they encounter substances with a certain pH value. For example, litmus, which is a natural indicator, turns red when it contacts something acidic and blue with alkaline materials. Often, indicators are mixed to create something called a universal indicator, which can change to several different colors to indicate a wide range of acidity; usually from pH2-10. Most pH paper strips are impregnated with universal indicators.
Handle With Care
Handle With Care
PH papers often have a high fabric content. Sometimes they are more fabric than paper, although they are usually still called papers. Before the paper can be saturated, however, the universal solution must be created. Weak acids are mixed in a natural material container (not metal, which can react to the acid) and pure, distilled water. Commercial solutions usually contain PROPAN-2-OL, which is a flammable material and must be handled with care, in a well-ventilated area. Commercial universal pH solutions also contain IVN RAT LD50 (1088 mg/kg), ORL MUS LD50 (3600 mg/kg) ORL RAT LD50 (5045 mg/kg) and SCU MUS LDLO (6 gm/kg). Although these acids can be ordered online from places like ScienceLab.com, they should only be handled by a professional scientist.
Once the paper has been immersed in the solution and is thoroughly saturated, it can then be immediately removed and set out to dry. The drying area must be free of any chemicals or vapors. Once the paper has dried, it is usually cut into strips and stored in a tight container.
Homemade PH Paper
Homemade PH Paper
PH paper can be also made at home using natural ingredients. Before you begin, make sure you have an earthenware bowl to submerge your paper in, and also some distilled water. Use paper with a high fiber content.
Next, create an infusion of red cabbage leaves by boiling them in distilled water. Strain the leaves and slowly heat the liquid again until a third of it has evaporated. Place this liquid in the earthenware bowl, and submerge your paper in the bowl. Once saturated, remove the paper and dry it. The best way to dry it is to gently drape it over a string or bar. The paper will be a gray color. If it comes into contact with acid, it will turn red; alkalies will turn it green.
Cite This Article
MLA
Sanders, April. "How Is PH Paper Made?" sciencing.com, https://www.sciencing.com/ph-paper-made-4676070/. 24 April 2017.
APA
Sanders, April. (2017, April 24). How Is PH Paper Made?. sciencing.com. Retrieved from https://www.sciencing.com/ph-paper-made-4676070/
Chicago
Sanders, April. How Is PH Paper Made? last modified March 24, 2022. https://www.sciencing.com/ph-paper-made-4676070/