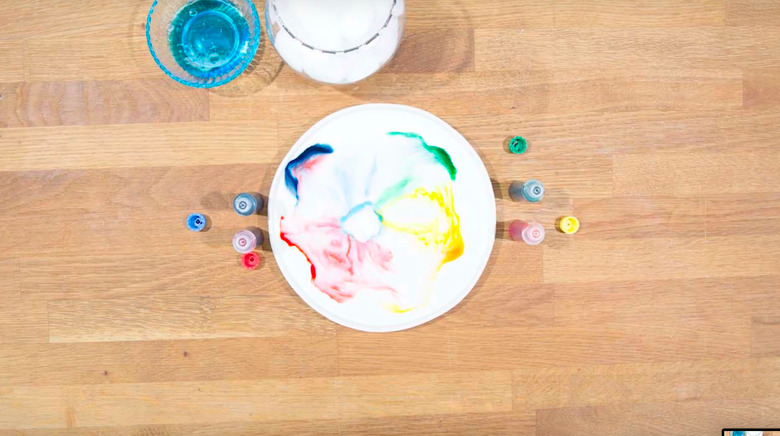Science Projects Using Dawn Dishwashing Liquid
Dawn dishwashing soap can be an eye-opening addition to most science classes. There are a variety of experiments or demonstrations that can be done using Dawn. Of course, it could also be interesting to try the same experiments with other dishwashing soaps and discover if the outcomes are the same or different.
Milk Experiment
Milk Experiment
For this demonstration, the teacher will need four different colors of liquid food coloring, a heavy paper plate, a cotton swab and whole milk.
1. Pour Milk on Paper Plate
Then place one drop of each food coloring toward the center of the plate, but the drops should not touch.
Next, ask the class what may happen if a drop of Dawn is placed on the cotton swab and then placed in the center of the plate.
2. Add Cotton Swab to Plate
After hearing some of the ideas, complete the experiment by placing a cotton swab with a drop of Dawn on the end in the center of the plate and prepare for a burst of color.
The fat from the milk bonds with the dishwashing liquid, pushing all the water content of the milk (and the food coloring) to the outside of the plate.
Density Experiment
Density Experiment
You will need eight ounces of each of these liquids–Light Karo, water, vegetable oil, Dawn dishwashing liquid (the blue kind), rubbing alcohol, lamp oil and honey. It may be desirable to color some of the liquids; the only ones that may not accept coloring would be the vegetable oil and the honey. Pour one liquid at a time in the center of a large, clear, glass container. It is important that the liquid does not touch the side. Continue until all the liquids are poured into the container. When they settle, there should be seven definite layers of liquids. At this point the density of the liquids could be discussed and why the ones at the bottom and top are located where they are.
Citrus Fizz
Citrus Fizz
This demonstration shows how citrus acid, sodium bicarbonate and Dawn dishwashing soap can work together to create a fizzy, foaming creation. First, a spoonful of baking soda should be poured into a glass, then place in the glass a squirt of Dawn. After this, quarter a lemon and squeeze the juice from one of the quarters of lemon into the glass. Stir this mixture and watch the chemistry take over.
Dry Ice
Dry Ice
Perform this experiment with care. Dry ice should never touch exposed skin; it can cause very bad burns, so wear thick gloves or use tongs when handling it. Fill a tall glass with warm water, then add a squirt of Dawn. Using the gloves or tongs, carefully place a piece of dry ice into the container next and see how it reacts. The soap in the water traps the carbon dioxide and water vapor that is being let off by the dry ice, and forms bubbles instead of a dry ice cloud that is generally given off by the dry ice.
Cite This Article
MLA
Napier, Kimberly. "Science Projects Using Dawn Dishwashing Liquid" sciencing.com, https://www.sciencing.com/science-using-dawn-dishwashing-liquid-7969343/. 17 May 2019.
APA
Napier, Kimberly. (2019, May 17). Science Projects Using Dawn Dishwashing Liquid. sciencing.com. Retrieved from https://www.sciencing.com/science-using-dawn-dishwashing-liquid-7969343/
Chicago
Napier, Kimberly. Science Projects Using Dawn Dishwashing Liquid last modified August 30, 2022. https://www.sciencing.com/science-using-dawn-dishwashing-liquid-7969343/