One of the simplest ways to understand the structures and functions of the organelles housed within a cell – and cell biology as a whole – is to compare them with real world things.
For example, it makes sense to describe the Golgi apparatus as a packing plant or post office because its role is to receive, modify, sort and ship out cell cargo.
The Golgi body’s neighbor organelle, the endoplasmic reticulum, is best understood as the cell’s manufacturing plant. This organelle factory builds the biomolecules required for all life processes. These include proteins and lipids.
You probably already know how important membranes are for eukaryotic cells; the endoplasmic reticulum, which includes both the rough endoplasmic reticulum and smooth endoplasmic reticulum, takes up over half of the membrane real estate in animal cells.
It would be hard to exaggerate just how important this membranous, biomolecule-building organelle is to the cell.
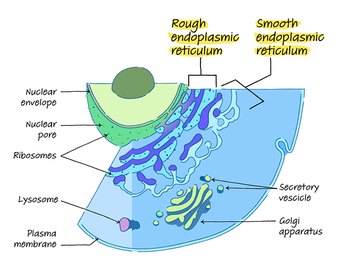
Structure of the Endoplasmic Reticulum
The first scientists to observe the endoplasmic reticulum – while taking the first electron micrograph of a cell – were struck by the endoplasmic reticulum’s appearance.
For Albert Claude, Ernest Fullman and Keith Porter, the organelle looked “lace like” because of its folds and empty spaces. Modern observers are more likely to describe the endoplasmic reticulum’s appearance as like a folded ribbon or even a ribbon candy.
This unique structure ensures that the endoplasmic reticulum can perform its important roles within the cell. The endoplasmic reticulum is best understood as a long phospholipid membrane folded back on itself to create its characteristic maze-like structure.
Another way of thinking about the endoplasmic reticulum’s structure is as a network of flat pouches and tubes connected by a single membrane.
This folded phospholipid membrane forms bends called cisternae. These flat discs of phospholipid membrane appear stacked together when looking at a cross section of the endoplasmic reticulum under a powerful microscope.
The seemingly empty spaces between these pouches are just as important as the membrane itself.
These areas are called the lumen. The internal spaces that make up the lumen are full of fluid and, thanks to way the folding increases the organelle’s overall surface area, actually make up about 10 percent of the cell’s total volume.
Two Kinds of ER
The endoplasmic reticulum contains two main sections, named for their appearance: the rough endoplasmic reticulum and the smooth endoplasmic reticulum.
The structure of these areas of the organelle reflect their special roles within the cell. Under a microscope’s lens, the phospholipid membrane of the rough endoplasmic membrane appears covered in dots or bumps.
These are ribosomes, which give the rough endoplasmic reticulum a bumpy or rough texture (and hence its name).
These ribosomes are actually separate organelles from the endoplasmic reticulum. Large numbers (up to millions!) of them localize at the rough endoplasmic reticulum’s surface because they are vital for its job, which is protein synthesis. The RER exists as stacked sheets that twist together, with helix-shaped edges.
The other side of the endoplasmic reticulum – the smooth endoplasmic reticulum – looks quite different.
While this section of the organelle still contains the folded, maze-like cisternae and fluid-filled lumen, the surface of this side of the phospholipid membrane appears smooth or sleek because the smooth endoplasmic reticulum does not contain ribosomes.
This portion of the endoplasmic reticulum synthesizes lipids rather than proteins, so it does not require ribosomes.
The Rough Endoplasmic Reticulum (Rough ER)
The rough endoplasmic reticulum, or RER, gets its name from its characteristic rough or studded appearance thanks to the ribosomes that cover its surface.
Remember that the whole endoplasmic reticulum acts like a manufacturing plant for the biomolecules necessary for life, such as proteins and lipids. The RER is the section of the factory dedicating to producing only proteins.
Some of the proteins produced in the RER will remain in the endoplasmic reticulum forever.
For this reason, scientists call these proteins resident proteins. Other proteins will undergo modification, sorting and shipping to other areas of the cell. However, a large number of the proteins built in the RER are labeled for secretion from the cell.
This means that after modification and sorting, these secretory proteins will travel via vesicle transporter through the cell membrane for jobs outside the cell.
The location of the RER within the cell is also important for its function.
The RER is right next door to the nucleus of the cell. In fact, the phospholipid membrane of the endoplasmic reticulum actually hooks up with the membrane barrier that surrounds the nucleus, called the nuclear envelope or nuclear membrane.
This tight arrangement ensures that the RER receives the genetic information it requires to build proteins directly from the nucleus.
It also makes it possible for the RER to signal the nucleus when protein building or protein folding goes awry. Thanks to its close proximity, the rough endoplasmic reticulum can simply shoot a message to the nucleus to slow down production while the RER catches up with the backlog.
Protein Synthesis in the Rough ER
Protein synthesis generally works like this: The nucleus of every cell contains a full set of DNA.
This DNA is like the blueprint the cell can use to build molecules like proteins. The cell transfers the genetic information necessary for building a single protein from the nucleus to the ribosomes at the surface of the RER. Scientists call this process transcription because the cell transcribes, or copies, this information from the original DNA using messengers.
The ribosomes attached to the RER receive the messengers carrying the transcribed code and use that information to make a chain of specific amino acids.
This step is called translation because the ribosomes read the data code on the messenger and use it to decide the order of the amino acids in the chain they build.
These strings of amino acids are the basic units of proteins. Eventually, those chains will fold into functional proteins and maybe even receive labels or modifications to help them do their jobs.
Protein Folding in the Rough ER
Protein folding generally happens in the interior of the RER.
This step gives the proteins a unique three-dimensional shape, called its conformation. Protein folding is crucial because many proteins interact with other molecules using their unique shape to connect like a key fitting into a lock.
Misfolded proteins may not function properly, and this malfunction can even cause human disease.
For example, researchers now believe that problems with protein folding may cause health disorders such as type 2 diabetes, cystic fibrosis, sickle cell disease and neurodegenerative problems like Alzheimer’s disease and Parkinson’s disease.
Enzymes are a class of proteins that make chemical reactions possible in the cell, including those processes involved in metabolism, which is the way the cell accesses energy.
Lysosomal enzymes help the cell break down unwanted cell contents, such as old organelles and misfolded proteins, in order to repair the cell and tap the waste material for its energy.
Membrane proteins and signaling proteins help cells communicate and work together. Some tissues need small numbers of proteins while other tissues require a lot. These tissues usually dedicate more space to the RER than other tissues with lower protein-synthesis needs.
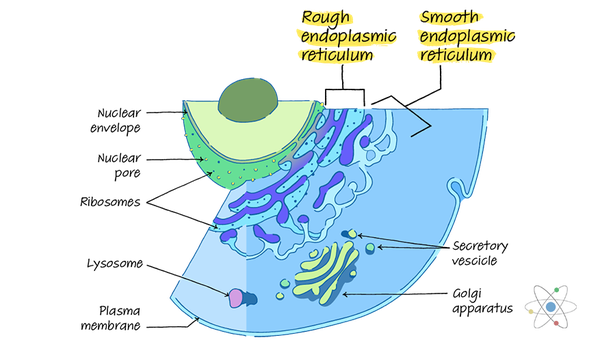
The Smooth Endoplasmic Reticulum (Smooth ER)
The smooth endoplasmic reticulum, or SER, lacks ribosomes, so its membranes look like smooth or sleek tubules under the microscope.
This makes sense because this portion of the endoplasmic reticulum builds lipids, or fats, rather than proteins, and thus doesn't need ribosomes. These lipids may include fatty acids, phospholipids and cholesterol molecules.
Phospholipids and cholesterol are needed for building plasma membranes in the cell.
The SER produces lipid hormones that are necessary for the proper functioning of the endocrine system.
These include steroid hormones made from cholesterol, such as estrogen and testosterone. Because of the major role the SER plays in hormone production, cells that require lots of steroid hormones, like those in the testes and ovaries, tend to dedicate more cellular real estate to the SER.
The SER is also involved in metabolism and detoxification. Both of these processes happen in liver cells, so liver tissues usually have a greater abundance of SER.
When hormones signals indicate that the energy stores are low, kidney and liver cells begin an energy-producing pathway called gluconeogenesis.
This process creates the important energy source glucose from non-carbohydrate sources in the cell. The SER in liver cells also helps those liver cells remove toxins. To do this, the SER digests portions of the dangerous compound to make it water soluble so that the body can excrete the toxin through the urine.
The Sarcoplasmic Reticulum in Muscle Cells
A highly specialized form of the endoplasmic reticulum shows up in some muscle cells, called myocytes. This form, called the sarcoplasmic reticulum, is usually found in cardiac (heart) and skeletal muscle cells.
In these cells, the organelle manages the balance of calcium ions the cells use to relax and contract the muscle fibers. Stored calcium ions absorb into the muscle cells while the cells are relaxed and release out of the muscle cells during muscle contraction. Problems with the sarcoplasmic reticulum can lead to serious medical problems, including heart failure.
The Unfolded Protein Response
You already know that the endoplasmic reticulum is a part of protein synthesis and folding.
Proper protein folding is crucial for making proteins that can do their jobs correctly, and as previously mentioned, misfolding can cause proteins to function improperly or not work at all, possibly leading to serious medical conditions such as type 2 diabetes.
For this reason, the endoplasmic reticulum must ensure that only correctly folded proteins transport from the endoplasmic reticulum to the Golgi apparatus for packaging and shipping.
The endoplasmic reticulum ensures protein quality control through a mechanism called the unfolded protein response, or UPR.
This is basically very rapid cell signaling that enables the RER to communicate with the cell nucleus. When unfolded or misfolded proteins begin to pile up in the lumen of the endoplasmic reticulum, the RER triggers the unfolded protein response. This does three things:
- It signals the nucleus to slow down the rate of protein synthesis by limiting the number of messenger molecules sent out to the ribosomes for translation.
- The unfolded protein response also increases the endoplasmic reticulum’s ability to fold proteins and degrade misfolded proteins.
- If neither of these steps solve the protein pile up, the unfolded protein response also contains a failsafe. If all else fails, the affected cells will self destruct. This is programmed cell death, also called apoptosis, and is the last option the cell has to minimize any damage unfolded or misfolded proteins could cause.
ER Shape
The shape of the ER relates to its functions and can change as needed.
For example, increasing the layers of RER sheets helps some cells secrete greater numbers of proteins. Conversely, cells such as neurons and muscle cells that do not secrete as many proteins may have more SER tubules.
The peripheral ER, which is the portion not connected with the nuclear envelope, can even translocate as needed.
This reasons and mechanisms for this are the subject of research. It may include sliding SER tubules along the microtubules of the cytoskeleton, dragging the ER behind other organelles and even rings of ER tubules that move around the cell like small motors.
The shape of the ER also changes during some cell processes, such as mitosis.
Scientists are still studying how these changes take place. A complement of proteins maintain the overall shape of the ER organelle, including stabilizing its sheets and tubules and helping determine the relative amounts of RER and SER in a particular cell.
This is an important area of study for researchers interested in the relationship between the ER and disease.
ER and Human Disease
Protein misfolding and ER stress, including stress from frequent UPR activation, may contribute to human disease development. These may include cystic fibrosis, type 2 diabetes, Alzheimer's disease and spastic paraplegia.
Viruses may also hijack the ER and use the protein-building machinery to churn out viral proteins.
This can alter the shape of the ER and prevent it from performing its normal functions for the cell. Some viruses, such as dengue and SARS, make protective double-membraned vesicles inside the ER membrane.
References
About the Author
Melissa Mayer is an eclectic science writer with experience in the fields of molecular biology, proteomics, genomics, microbiology, biobanking and food science. In the niche of science and medical writing, her work includes five years with Thermo Scientific (Accelerating Science blogs), SomaLogic, Mental Floss, the Society for Neuroscience and Healthline. She has also served as interim associate editor for a glossy trade magazine read by pathologists, Clinical Lab Products, and wrote a non-fiction YA book (Coping with Date Rape and Acquaintance Rape). She has two books forthcoming covering the neuroscience of mental health.